Zoom: https://ethz.zoom.us/j/66776813667
Interface stability in all-solid-state batteries
Corsin Battaglia (Materials for Energy Conversion – EMPA Dübendorf)
All-solid-state batteries combining an alkali metal anode and a high-voltage cathode have the potential to double the energy density of current-generation rechargeable batteries. However, interface stability remains a major challenge. On the anode side, alkali metal dendrites penetrating into the solid electrolyte limit the maximum plating current density and prevent fast charging, while on the cathode side the limited oxidative stability of solid electrolytes is a major challenge, especially when the battery is charged beyond 4V.
We recently discovered that the critical current density for dendrite formation in the archetypical ceramic solid electrolyte Na-β”-alumina can reach 10 mA/cm2 at room temperature, which is ten times higher than that measured on a garnet-type Li7La3Zr2O12 electrolyte [1]. Further, we demonstrated that above the melting temperature of sodium, a cumulative capacity of >10 Ah of sodium can be plated and stripped at an unprecedented current density of 1000 mA/cm2 without dendrite formation [2] indicating that the alkali metal and not the electrolyte prevent fast charging at room temperature.
We recently also demonstrated the integration of hydroborate electrolytes with a 4 V class cathode through in-situ formation of a passivating interface layer [3]. Combined with their high ionic conductivity >1 mS/cm at room temperature, low gravimetric density 1.2 g/cm3, low toxicity, high thermal and chemical stability, stability vs lithium and sodium metal, soft mechanical properties enabling cold pressing, compatibility with solution infiltration, and potential for low cost, hydroborate electrolytes represent a promising option for a competitive next-generation all-solid-state battery technology [4].
____
[1] M.-C. Bay, M. Wang, R. Grissa, M. V. F. Heinz, J. Sakamoto, C. Battaglia, Adv. Energy Mater. 2019, 201902889
[2] D. Landmann, G. Graeber, M. V. F. Heinz, S. Haussener, C. Battaglia, Materials Today Energy 2020, 18,
[3] R. Asakura, D. Reber, L. Duchêne, A. Remhof, H. Hagemann, C. Battaglia, Energy Environ. Science 2020, 13, 5048
[4] L. Duchêne, A. Remhof, H. Hagemann, C. Battaglia, Energy Storage Mater. 2020, 25, 782
Sculpting hydrogels using advective processing
Alexandra Bayles (Soft Materials – D-MATL)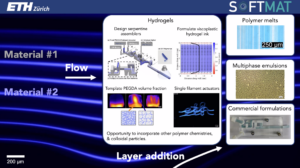
Polymeric hydrogels, water-laden 3D crosslinked networks, find broad application as advanced biomaterials and functional materials due to their biocompatibility, stimuli responsiveness, and affordability. In these materials, the crosslinking density reports material properties such as elasticity, strength, permeability, and swelling propensity. Patterning this critical design parameter across the volume polymerized is an attractive means by which to engineer hydrogel performance. In this talk, we present a novel processing scheme that uses laminar flow to direct the organization of hydrogel crosslinking density across a single sample. Inspired by techniques used to structure polymeric melts, we design custom millifluidic devices that force disparate streams through serpentine splitting, rotation, and recombination elements. These elements multiply the advecting macromer concentration distribution within the cross-sectional area while preserving its relative spacing and orientation. Incorporating advective assembly devices into conventional 3D printing nozzles enables the fabrication of hierarchical, shape-morphing hydrogels. This work exemplifies advective processing’s potential to encode soft material microstructure and subsequently functionality through geometrically dictated, generalizable flows.

